- Blog
- Comenity bank credit cards victoria secret
- Minecraft steve memes
- Dreambox hackes
- Lift equifax freeze without pin
- File shredder windows 10 free
- Popular sans serif typeface
- National grid gas plan types
- Microsoft office professional 2007 product key free
- Download hindi audio tracks for hollywood movies
- Shortcuts in powerpoint for bullet point
- Logarithmic scale decibels
- Solving quadratic equations by factor practice questions
- Among us halloween coloring page
- Ms office 2013 free download filehippo
- Content control in microsoft word
- Download bluestacks android emulator on your pc
- Minecraft pe 0-13-0 apk free download
- Best video editing app for iphone youtube
- Idm download full version with crack free
- Fantasy forest story mod apk download
- Free serial number for vegas pro 13
- Pokemon x and y gba download zip
- Picasa web album archive
- Pixelmon mod 1-12-2 maps
- Concrete screen blocks near me
- Steve minion butt 125 wow
- Sandisk usb flash repair tool
- Weight tracker template with graph
- Walkthrough superpowered
- Couples bang the babysitter cumshot porn
- Eva padlock boobs
- Free spider solitaire games without ads
- Solving quadratic equation by factoring
- Registration code fight night champion pc
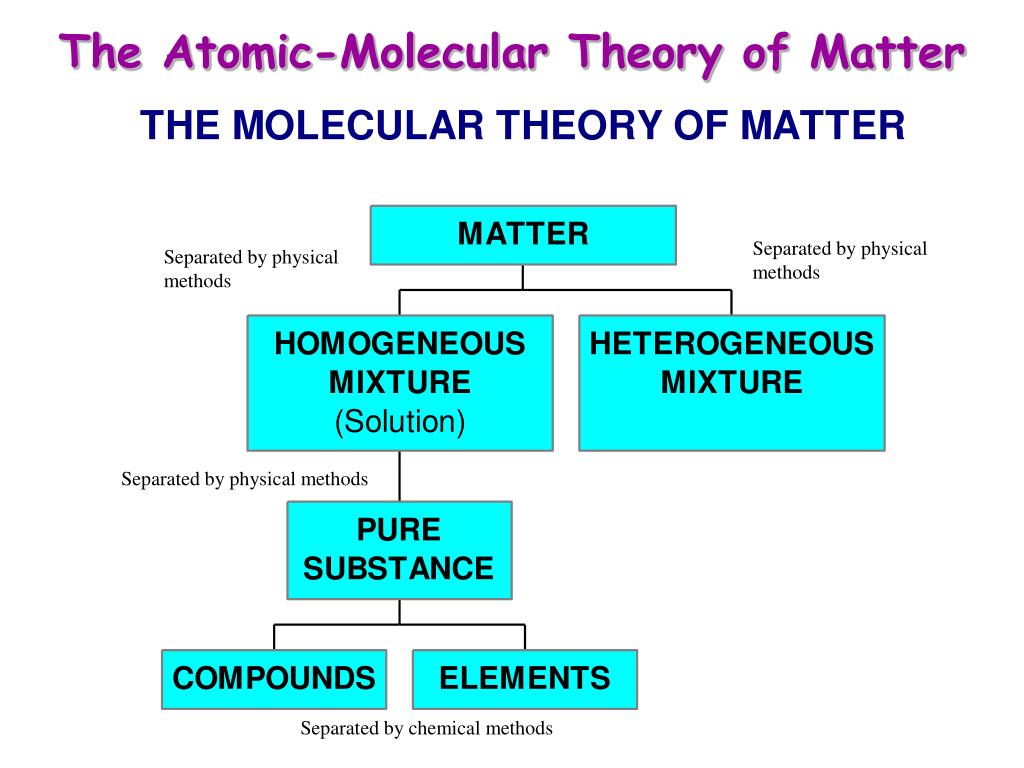
- Atomic molecular theory
- Blank brochure design templates free download
- Calculate volume of tank

When an atom contains more or fewer electrons than protons, it has a negative or positive overall charge, and these atoms are known as ions.Īn atom is exceedingly small, much smaller than our imagination allows us to imagine.
Like the layers of an anion, these electrons are organised in orbits around the nucleus of the atom. To have zero charge, an element's atom must have the same amount of protons as electrons. A single negative charge is carried by an electron. The total number of protons and neutrons in the nucleus is used to calculate an element's atomic weight. The number of protons or positive charges in the nucleus determines an element's atomic number. A proton is a particle with a single positive charge and a mass of one unit. A neutron is a neutral particle with a mass of one unit. The nucleus is made up of neutrons and protons, which are responsible for an atom's weight and positive charges. The atom is electrically neutral if the number of protons and electrons is equal.
/GettyImages-1003408458-333023afaac44d33bd3d94123b95f8f8.jpg)
Protons have a positive electric charge, while electrons have a negative charge and neutrons don't have any. The nucleus contains more than 99.94% of an atom's mass. Only one type of hydrogen, the most prevalent, lacks neutrons.

One or more protons and a number of neutrons make up the nucleus. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail.Ītoms Molecules and Ions Atoms and Molecules DefinitionĮvery atom is made up of a nucleus and one or more electrons attached to it. Atoms are extremely small, measuring about 100 picometers in diameter. Every solid, liquid, gas, and plasma is made up of neutral or ionised atoms. This results in an increase in the kinetic energy of the particles.An atom is the smallest unit of matter that makes up a chemical element. As the temperature of a sample of gas is increased, the speeds of the particles are increased.
